Call me a geek, but I can’t help thinking of my school science lessons every time I take a shower. It’s the problem of the mixer taps. What actually de we mean by temperature? Isn’t it just the heat? If the day is hot, you feel the heat – what’s the problem? Back then, as a 14-year old it all seemed so confusing. In a science practical we once had to make a thermometer out of glass tubing and suck up some red liquid.
I distinctly remember the ever-patient Mr Sills trying to explain the strange word “calibration” as we plunged our glass-tubes into bowls of ice, scratching marks on the glass as we did so.
Many decades later I can now see there’s a fundamental difficulty caused by our confusing language. ‘Hot’ is an adjective, ‘heat’ a noun. It’s a pity they sound so similar, given they play different roles in language and have such distinct meanings. Then there’s the opposite; ‘cold’ is the adjective and …… what is the noun: coldth? coldness? Unfortunately, we tend to use the same word for both: “It’s cold in here. Shut the window, you’ll let the cold in” – ambiguity in language hampering our efforts to understand.
The confusion partly dates back to a discredited theory, popular in the eighteenth century Back then, long before the advent of steam locomotives and central heating, scientific efforts to understand heat gave rise to the idea that it was a kind of fluid. After all, it flowed along teaspoons from the hot end to the cold, just like rainwater, flowing from the higher to the lower end of a gutter. Steeped as science was in the classics, this supposed liquid was named ‘caloric’, after the Latin word for heat.
The development of the new science of thermodynamics in the mid nineteenth century was driven by the arrival of steam engines, used originally to pump water out of mines which tended to flood as they were dug ever deeper. The new technology inspired new science which resulted in an altogether different and powerful concept of heat. This is broadly how we understand it to this day. It isn’t any kind of extraneous thing added into stuff, not a fluid running through the metal of a teaspoon. It’s simply an intrinsic property of the stuff of the teaspoon itself. Fortunately, scientific understanding of the nature of ‘stuff’ was also developing around the same time. This led to the concept of what we now know as atoms and molecules (groups of atoms bonded together). All materials – solid liquid and gaseous – are composed of these.

What was soon realised is that these atoms and molecules aren’t just sitting quietly together, they are incessantly moving. They may be dashing about freely and separately like midges in a summer’s day – that’s how it is in gases; or they may be more closely packed, sliding past each other, like balls in a ball-pit – that’s the liquid state.
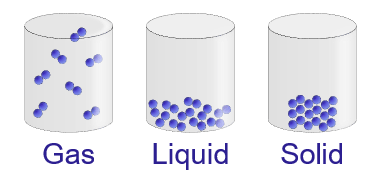
If they are simply sitting next to one another, as in a solid, they are still in active motion, this time in the form of vibration around a fixed point. Heat, it was soon realised, is simply the net effect of all this movement, summed up over the zillions of separate atoms or molecules that make up any piece of matter.
With this more enlightened idea – all the atom or molecules in a thing jiggling or shifting around – comes greater clarity. If they are moving, they must have energy – the faster they move , the greater the energy. “Heat” is simply the energy they have due to their motion. The faster they move, the greater the energy. In fact scientists tend to call it “internal energy” rather than heat. The “heat” in a bath or a room or a star simply means the sum total of all the energy of all the atoms and molecules moving in a thing. As a bath fills up, more and more are being added, each with its own energy of motion; so the total amount of heat (or internal energy) is increasing. And you’re paying for it through your energy bills.
But there’s another catch – your bath could be warm or hot. You’re paying not only for the quantity of hot water you use, but the ‘quality’ of it too – what we call the temperature. High temperature heat is high quality heat. The temperature reflects how fast the atoms and molecules are moving, on average. The faster the molecules are moving, the greater their energy of motion and this we experience as a higher temperature.
A spark from a sparkler is obviously “hotter” than a warm bath – it could burn you. The molecules in a warm bath are many but moving relatively slowly, on average; those in a spark are much fewer, but they’re jiggling around much more quickly.
So there we have it – the basic concept of thermodynamics. Heat is a form of energy (more correctly called internal energy) and it represents the total energy of all the molecules making up a thing. Temperature, on the other hand, is a measure of the ‘quality’ of that energy – high or low. It represents the average speed of all the molecules. A bath contains a lot of low quality heat; a sparkler a little bit of high quality heat.
So when you take a shower, you balance water from the hot and cold taps to give you the right temperature. If the temperature is too low, you can increase the proportion of high energy molecules by opening the hot tap, or you can reduce the proportion of lower energy ones by closing the cold tap. Either way you’ll raising the average energy of the molecules, thus raising the temperature.
Further reading
Heat on the move
Now, molecules don’t hang around as singletons, they stick together in their zillions, especially in the solid or liquid form. So, what happens when a group of molecules with high energy sit cheek by jowl with some less energetic neighbouring ones? As you can imagine, the vibrations or shifting motion of the one will gradually transfer to the other. Energy inevitably moves from the high energy molecules to the surrounding lower energy ones – and certainly not the other way round.
‘Heat’ is simply the sum total of all the energy of motion in all the molecules in a thing. So, when heat flows along a teaspoon from a hot cup of tea to your hand, which is cooler, energy is passed along from the more energetic molecules in the tea to the less energetic ones in your hand.
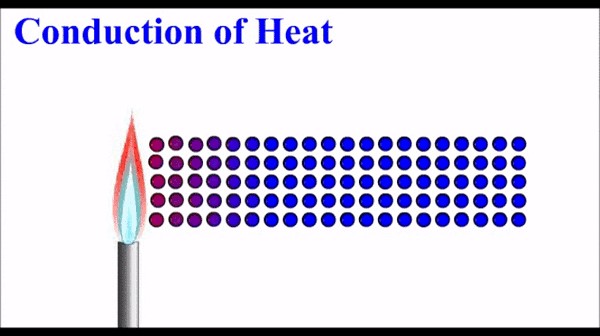
It’s not anything concrete that moves along the teaspoon – not the molecules themselves, nor some imaginary ‘caloric’ fluid flowing between them, but simply the energy of movement passing from more agitated molecules to less agitated ones. Click here to see a nice animation of the diagram
It’s because what we experience as heat is really just a manifestation of the movement of molecules inside matter that scientists prefer the phrase ‘internal energy’ to ‘heat’ – after all that’s what it really is.
So, to nail this concept of ‘heat’ or internal energy, let’s look more closely at some homely examples of it. Your shower is pouring out hot water at a steady rate. Zillions of water molecules are rushing past, each carrying their bit of internal energy (or heat). You are paying for this through the gas flames or electrical heater that gave internal energy to your hot water in the first place. When you set your thermostat you are deciding how fast these molecules will be moving on average – the temperature. The higher the temperature, the faster they move, the more energy you consume, the more you pay. Equally when you open up the hot tap, the faster the flow of these molecules. Again you pay more, not because the molecules are moving faster, but because more of them are passing each second; you pay for both quality and quantity.
Now let’s take a hot cup of tea. The molecules in it will be moving with a range of speeds, but on average they will have greater energy than those in a cool teaspoon placed in the cup. As you can imagine, the greater energy of the molecules in the hot tea will gradually transfer to the molecules in the spoon, passing from one agitated molecule to the next – like ripples on a pond. This elevated level of energy will spread along the spoon to your hand holding it. Of course it only spreads from the more energetic tea to the less energetic spoon – not the other way round! It’s easy to see how this corresponds to what we know intuitively about temperature – ‘heat’ will always pass from a higher temperature to a cooler one.
These simple ideas about heat and temperature help with understanding many aspects of everyday life. For example, when we insulate our homes to reduce heat loss, we are impeding the flow of energy through brick or glass caused by the difference of temperature between the inside and outside. When we close the door of a fridge, we are creating an enclosed box from which internal energy (or ‘heat’) in the food, air and walls can be extracted. Because this energy has to be taken from a cooler place (the fridge) to warmer one (the kitchen) it has to be pumped our – rather like the pumping needed to get water from the bottom of a mine to the top. Interesting stories of how heat energy moves around the home (remember “conduction, convection and radiation” from school?) and how it is absorbed during evaporation or melting will no doubt feature in future blogs.
© Andrew Morris 15th August 2020
To receive an email alert when each new blog is published, contact andrewmorris110@gmail.com to join the mailing list.
