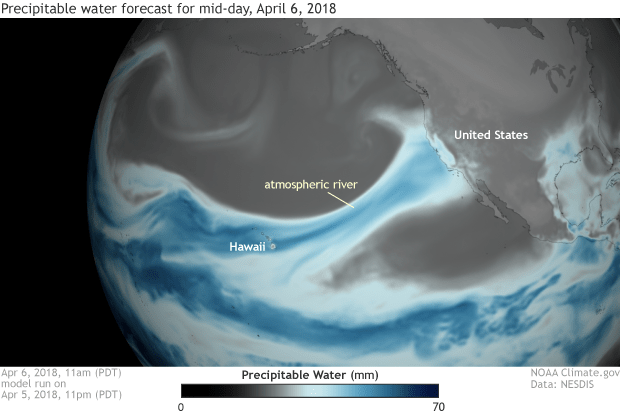
This dramatic image produced by the US National Weather Service shows a special weather event destined to bring huge rain storms to California. You can clearly see the “river” of high humidity air streaming across from Hawaii to California – known as the “Pineapple Express”
One of our readers in the USA asked if we could explore these so-called “atmospheric rivers”. One had a devastating impact in California in February this year. It makes a good starting point for exploring the many important scientific concepts underpinning why it rains so bloomin’ much (sometimes, in some places, at least).
The image created above uses colours to indicate how much water vapour is locked up in the atmosphere at every point. The darker blue colours show how warm conditions over Hawaii enable large quantities of water to be absorbed into the atmosphere and then travel with the wind.
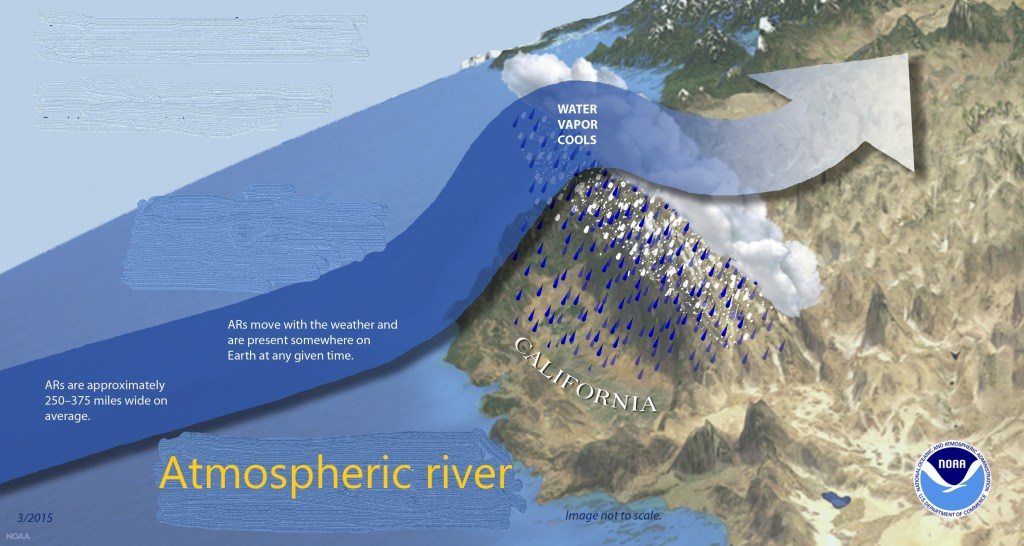
When winds drive that air eastwards it eventually makes landfall and meets the immovable object of the Sierra Nevada mountains. As this diagram shows, the air is forced upwards as it crosses the mountains and, in so doing, cools down (it’s colder up there!). The result is rain, or even snow and if conditions are bad the result can be extreme flooding down in the plain.
But, as this is a science blog, let’s dig a little deeper and ask what exactly we mean by water vapour. What indeed is a vapour? And why should it lead to rain when it cools? Answering these questions opens up a whole world of interesting pehnomena – clouds, mist, frost, dew, humidity, perspiration – even the problem of drying your clothes on a wet Wednesday.
Humidity
It’s a bit of a shocker at primary school to be told it’s not emptiness that surrounds us all, but air. The thought of air as a thing, tangible and weighty, contradicts everything a child has come to experience. You can’t touch it, feel it or see it; how can it be a thing? As we grow up and piece things together gradually, however, we do concede that wind must be a movement of air and that is on a blustery day it’s certainly weighty and only too tangible as it buffets around us and knocks over trees.
At some point we learn a little more about this invisible presence: that it’s in fact not a single kind of stuff but a mixture of several different things – nitrogen and oxygen mainly with a bit of carbon dioxide (increasingly so, unfortunately) we are told. We learn that these invisible things are all called gases and there, for the majority of people who don’t continue with science, it largely rests. Gases seem pretty mysterious and, judging by the smell of some of them, pretty dodgy too.
To get a more sophisticated idea of gases it’s worth trying to imagine them all as made up of molecules – tiny units of matter.
In contrast to solids and liquids, the molecules in gases are all far apart from each other. They are also not sitting still; they are rushing around freely at enormous speeds, roughly as fast as an aeroplane, occasionally bumping into one another or objects in their path, as this animation shows (click here to see animation).
The word ‘mixture’ is used in science to signify that the differing molecules, whether of nitrogen or oxygen or carbon dioxide, are interspersed randomly amongst one another – like the pieces in a fruit salad. None is attached to another; they move around, spacing themselves out evenly, on average, throughout the space they occupy.
What is less easy to imagine is that there is also an additional kind of molecule present in air: the familiar H2O molecule: water. Instinctively we think of water as a liquid, but, pressed to think it through, we know that it can also exist in other forms: as a solid in ice or as a vapour, in humid air. We know that a water left out in the open soon disappears, especially on a hot day – it invisibly enters the atmosphere. More precisely, H2O molecules, which are rushing around inside liquid water gradually fly off from the surface of the liquid and mingle with those of nitrogen and oxygen already in the air. We call this process evaporation – i.e. becoming vapour.
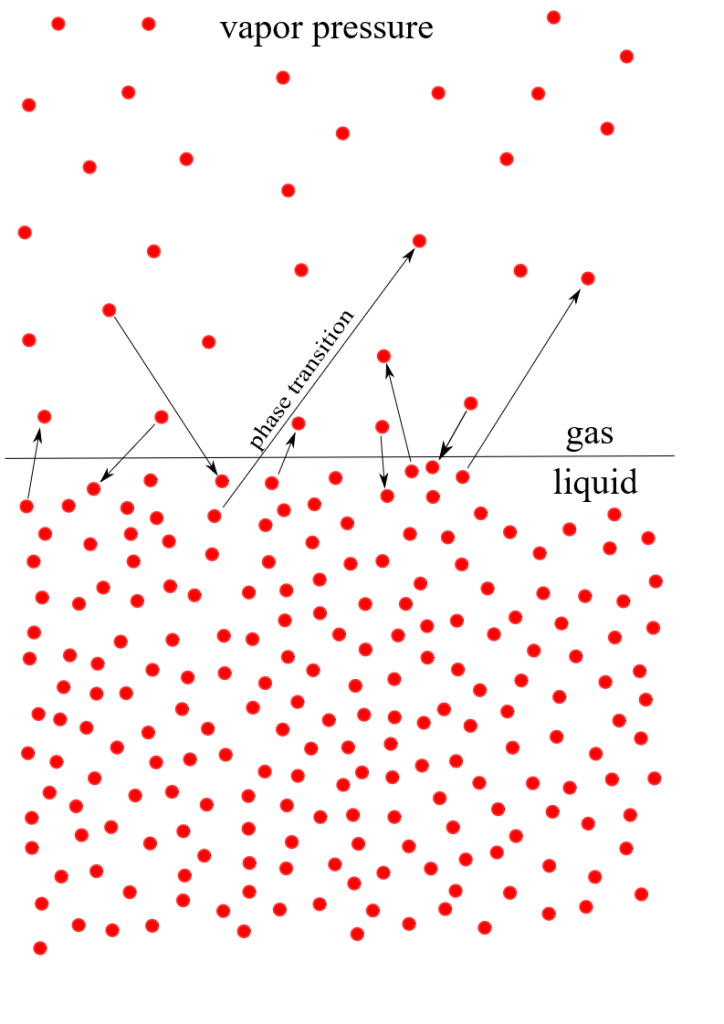
As you might imagine, this process of evaporation is happening all the time at water surfaces – molecules are leaving the top of a cup of tea, a bath, a reservoir or an ocean. The air is constantly filling up with H2O molecules. But it’s also losing them at the same time, as some molecules re-enter the liquid state from the air – a process we know as condensation.
You’re well aware of this when you boil and braise in the kitchen: you soon see the windows misting up with liquid water droplets – you’ve been busy evaporating water molecules. The same can occur in the confined space of a car: molecules of water vapour in your breath get absorbed into the air then condense out as a mist of liquid droplets on the windows.
Thinking one step further about your own experience, you’ll have noticed that this “steaming up” effect seems to occur more frequently when the air and windows are cold. It’s frosty outside, you start the car, the heaters haven’t yet done their work, your view soon gets blocked by water droplets condensing on the windscreen. Very frustrating. It’s the same when, out strolling on a bright frosty morning, your breath becomes visible as a cloud of water droplets as it hits the cold air.
Clouds of steam are perhaps most familiar when they puff out from the spout of a kettle of boiling water. On close examination we see that these clouds, like mist or fog, consist of a mass of tiny water droplets, suspended in the air: they are composed of liquid water, formed whenever the vapour hits cooler air. If you look closely at a boiling kettle you’ll see there is a small space next to the spout which is free of these steamy clouds. This is the invisible water vapour just before it cools down to form the visible stuff.
So, there we have it, water vapour, present everywhere in the air, is in fact an invisible gas. It’s just another one of the mixture of gases in air, alongside nitrogen and oxygen and carbon dioxide. The obvious question now is: why do we call it a vapour if it’s just a gas? Is there indeed any difference between these two words?
In common parlance we use the two words interchangeably, with vapour as perhaps the more poetic of the two. Shakespeare talks of “this most excellent canopy, the air….this majestical roof, fretted with golden fire, why it appears no other thing to me than a foul and pestilent congregation of vapours”. He doesn’t say “gases” and that’s no surprise: the word wasn’t invented till the 1650s, some time after he had died.
Scientists have come to exploit this fortunate duplication of words, to distinguish between two different conditions. During the nineteenth century it was discovered that if you compress a gas hard enough you can force it to turn into a liquid – to liquify it. Today, with cylinders of oxygen being delivered to hospitals for breathless patients and butane to country cottages for cooking, we have become familiar with the idea of “gases” being stored in liquid form, but only within very strong cylinders that can withstand the pressure.
It was soon found, however, that gases can only be liquefied when they are cooled below a certain temperature. Oxygen, for example must be below -119°C for this to occur. That’s why we don’t ordinarily see liquid oxygen hanging around. In fact, for familiar gases like nitrogen and hydrogen this so called critical temperature is very low indeed. The great exception is H2O. As we well know, water can be in the liquid state at any temperature below 100° C (and above 0°C) under normal conditions. That’s the temperature below which water vapour can condense into liquid water.
So under normal, everyday conditions, H2O can be in the liquid or vapour state whereas oxygen, nitrogen and carbon dioxide must remain gases. This leads to the simple distinction: gases are called vapours when they are at a temperature and pressure that enables them to be liquified. For H2O, this is the case at everyday temperatures and pressures. So, here on Earth where temperatures in temperate regions are typically around 20 – 40°C and pressures around one Bar, our air contains water as a liquid or vapour but nitrogen, hydrogen and oxygen only as gases.
This piece of Physics theory may seem rather abstract and complicated, but of course it’s the key to understanding how weather and climate work.
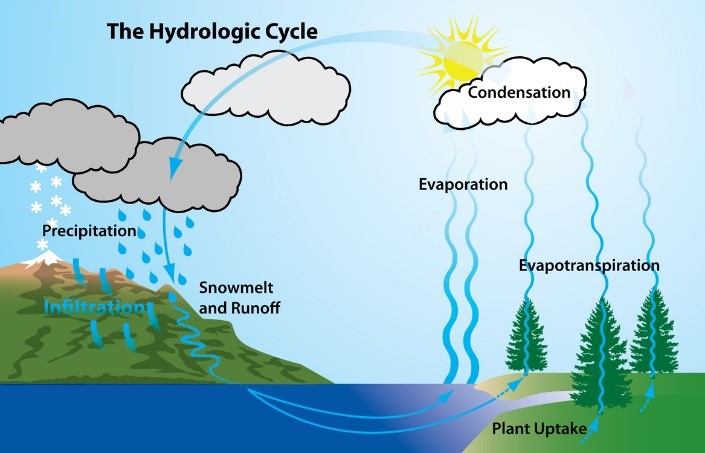
The ability of H2O molecules to cluster together as a liquid or space themselves apart as a vapour underpins the formation of rain, mist, clouds and fine weather. It enables water to cycle through its phases in the oceans and atmosphere, and, when it precipitates, in streams and rivers. All life depends on this marvellous roundabout.
One further nagging question remains before we get back to the “Pineapple Express” hitting California: what causes rain to form at all? Why doesn’t water vapour just stay as such, locked up invisibly in the atmosphere?
As the water cycle diagram shows, and residents of mountainous regions know, rain tends to fall mainly in the uplands. Air in the winds blowing across an ocean pick up water evaporating from the ocean. Saturated with water vapour, this moist air travels happily until it reaches hill country, such as the western coasts of Ireland and Scotland in the UK or the Sierra Nevada in the USA. Here the air is forced by the rising land to move upwards. The higher it goes the cooler it becomes, as any mountain climber will testify.
The final piece of theory helps explain what happens next. Between molecules in a substance there is a very slight force of attraction – this is what holds them together when they are in the solid form. However in a gas or vapour the molecules are moving so fast past each other that these forces have very little time to influence one another.
If they were to slow down, however, spending a little more time in close proximity to one another, this slight force of attraction is sufficient to bring them into contact with one another. They aggregate into what are in effect tiny regions of liquid water. This is the process we know as condensation (click here to see simulation)
Now, molecules move more slowly when the temperature is lower and faster when it is higher; that is effectively what temperature is – a reflection of the speed of molecules. When they move from a warmer to a cooler zone they slow down. This is exactly what happens as molecules in the air rise up mountainsides; they pass through ever cooler layers as they rise and consequently lose speed. At some point they will have slowed down enough for the forces between them to draw them together into clusters.
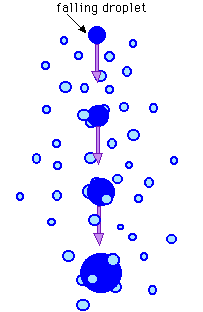
These tiny regions of liquid water gradually grow in to water droplets, ultimately forming regions of tiny droplets – what we see as clouds. When conditions are right, these droplets grow sufficiently large to fall under gravity as raindrops.
Image courtesy of Shodor

And if the temperature is especially low up there they can freeze together as ice. This is the origin of hail, snow and sleet – the magical consequence of water vapour in the atmosphere reaching the freezing cold conditions high up in the atmosphere.
Image courtesy of NASA
H2O molecules are amazingly versatile, not only able to shift from their vapour state to liquid (condensation), but also, in a further transformation, from liquid to solid (freezing). Once again it’s the force of attraction between the molecules that does the trick.
Returning to the plight of Californians (and plenty others around the world), beset with alternating floods, fires and droughts, the so called “atmospheric river” is simply a particular instance of the movement of air soaked in water vapour from warmer climes, hitting a cooler zone and discharging its load. The ability of the air mixture to welcome water vapour molecules where its warm but to lose them where its cooler is the very essence of the Earth’s beneficent cycle – transferring and desalinating ocean water to irrigate the parched land. Maintaining this life-support system depends critically on the precise balance of temperatures in each region – a crucial balance which humankind is sadly upsetting. Let us hope that some appreciation of the delicacy of this balance eventually descends on our political leaders.
© Andrew Morris 26th August 2019
