
“Why exactly (that is, scientifically) do we need paper, kindling then logs to get a fire started?”. Thus wrote one of our readers recently. It’s an interesting question arising from an everyday moment, chopping wood. I realised fire and flame had never played much of a part in my science education. A quick look in a chemistry text book and on the internet showed, it doesn’t feature much in standard texts either.
Yet, as our reader pointed out, fire is fast becoming a big global issue, given the recent conflagrations in Australia and the general threat posed by climate change.
There’s no great mystery about the subject. Our intuitions from watching fires flare up in the wind and die down when they are doused helps us grasp the basics. It’s clear that you need some kind of fuel and plentiful air around it. But, given that coal and wood don’t just burst spontaneously into flames under normal conditions, we are also aware of a vital third factor: a high enough temperature around the fuel to ignite it. Let’s take a look at these three factors, one by one.
Fuel
Wood, paper, coal, gas, oil – all share a key feature, from the chemical point of view. They are composed mainly of molecules that combine with oxygen, releasing energy in the process. Some everyday examples are:
Natural gas: a mixture of hydrocarbons, including methane (CH4)* and ethane (CH6)
(* CH4 is just a symbol for the methane molecule, comprising one carbon and four hydrogen atoms)
Petrol: a mixture of many hydrocarbons with names like isooctane, butane, 3-ethyltoluene
Wood: a complex structure composed of three main elements: cellulose, hemicellulose and lignin
Here are diagrams of some molecules in fuels: in gas, petrol and wood respectively. The presence of carbon atoms (black) and hydrogen (white) is common to all. Cellulose is a polymer made of units of glucose (a type of sugar) which contain oxygen atoms (red) as well.
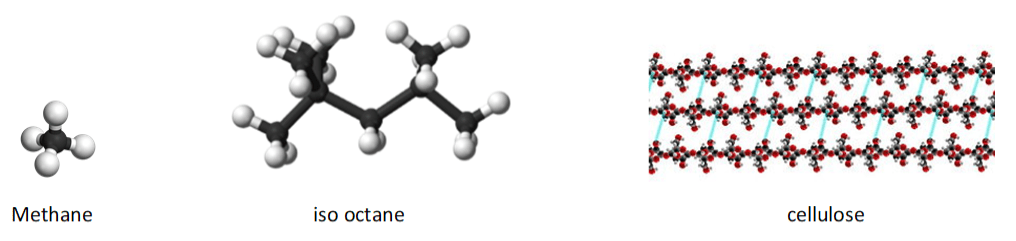
Wood
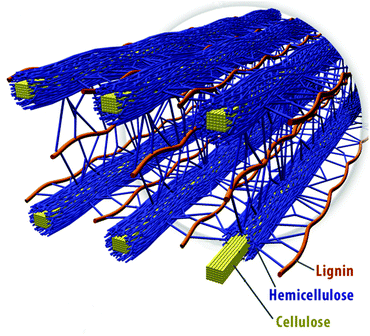
The key components of wood, from the point of view of combustion, are cellulose, hemicellulose and lignin. These are all long molecules built of repeating units (technically, polymers). They are arranged, as the diagram suggests, in a structure that ensures tensile strength (i.e. difficult to break lengthways) and also allows for the easy flow of nutrients. It’s the cellulose that ultimately fuels the fire.
The structure can be likened to reinforced concrete, with lignin playing the role of concrete and cellulose, the steel bars running through it.
Paper
Paper is made from wood by pulping it, a chemical process that separates the lignin from the cellulose. The pulp is then pressed and dried to produce paper, composed mainly of cellulose molecules. It is clearly highly combustible as it not only contains the energy yielding cellulose but has also lost its water content. This gives us one clue about why paper burns so easily that it’s often used to start a fire: the dryness of paper in comparison to wood.
Air
Air is a mixture of gases – mostly nitrogen (78%) and oxygen (21%) approximately. This means under open air conditions there is a plentiful supply of oxygen molecules to sustain the chemical reactions that we experience as fire.
High initial temperature
But as we know from considering an unlit fireplace, stocked up with newspaper pages and a stack of kindling wood, surrounded by air: nothing is going to happen spontaneously. A fire has to be ignited. A flaming matchstick against an edge of paper will do the trick or a slow burning “fire lighter”. It’s clear that somehow a high temperature environment must be created before the combustion process will begin.
Combustion
As we know from common experience there are distinct stages to combustion. At first, the initial temperature of the fuel has to be raised dramatically (to at least 350°C). The flame of a match or firelighter, a spark from a nearby fire or a flash of lightning can achieve this, as the recent bush fires in Australia demonstrate vividly. This elevated temperature causes the molecules of cellulose in the paper or wood to vibrate vigorously, ultimately breaking the bonds that hold the units of the molecular chain together.
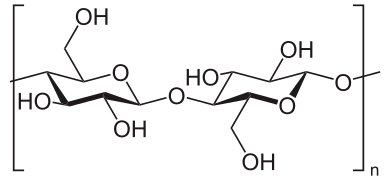
Cellulose molecules consist of long repeats of a simple unit comprising two linked molecules of glucose. The high temperature of the match flame break up the cellulose chain into smaller molecules.
Repeating unit of the cellulose molecule
In the second stage, these form flammable gases which rise into the surrounding air where they react with oxygen molecules in the air. These oxidation reactions release energy locked up in the bonds that previously held the separate oxygen and hydrocarbon molecules together. It is this energy which then goes on to ignite flames and heat up the surrounding fuel further, causing even more combustion. In this way the burning process becomes self-sustaining.
Products of combustion
The products of the reaction between the gases from the wood and oxygen in the air are carbon dioxide and water, if the combustion process is complete. However the more usual incomplete combustion process can produce a range of intermediate compounds that remain unburned because they fail to interact fully with the oxygen. These include carbon monoxide and other hydrocarbon gases (such as methane and benzene), some of which are toxic. It’s strange to think that water is created in a fire – it is in the invisible form of a vapour. You can see the effect of this when water vapour created in a central heating boiler condenses into steam and emerges via an external pipe.
As an aside, it’s interesting to note that the taking in oxygen and giving out water and carbon dioxide is reminiscent of the biological processes of breathing in and out and excretion. This is no coincidence! The cells in your body ‘burn’ food to produce energy, much as a fire does. But in the cells of your body it is a highly controlled process, regulated by enzymes, at a modest temperature of 37°C. Nevertheless, the release of energy by combining oxygen with hydrocarbons is the underlying chemical process in both cases.
Char
Incomplete combustion means some carbon is left behind as charcoal. A layer, known as ‘char’, develops on the outer surface of burning logs. However, the gases produced by burning inside the logs have to get away somehow. The char layer cracks as the wood shrinks allowing gases to flow out causing the familiar hissing sound and the jets of flame in a log fire. Any gas that fails to escape in this way builds up inside the log where the pressure may ultimately force small fragments of char to break off and spit out making the crackling sound we associate with a wood fire.
Smoke
As we’ve seen above, some of the gases created by burning logs may not be ignited as they mix with oxygen in the air – incomplete combustion. Molecules of these can condense into tiny droplets and form part of what we know as smoke, contributing to its characteristic smell. As many of these compounds are highly toxic, they can prove more lethal in fires than the heat of the fire itself. Smoke is a mist (technically an aerosol) comprising these liquid droplets and also solid particles – mainly carbon in the form of soot. The composition of smoke depends heavily on the temperature of the fire. The higher the temperature, the more complete the combustion and the fewer toxic compounds are present in the smoke.
Other products of combustion are more benign. Lignin, the other main component of wood, in addition to cellulose, is also transformed in the burning process, producing aromatic molecules which give smoked and barbecued food their flavours.
Ash

Ash, the solid material left after combustion is only around 1% or 2% of the mass of the original wood. It consists mainly of non-combustible minerals, typically calcium carbonate, some potash, and a little phosphate; as well as traces of elements such as iron and copper mainly as oxides. Gardeners sometimes use ash to enrich their soil with these compounds some of which helps neutralise acidity.
Starting a fire

So, returning to the questions that launched this little enquiry, what can we say about the need for paper and kindling? This image of the edge of piece of paper offers a clue. Cellulose has to be raised to around 350° C to start the combustion process (‘ignition’). The flame of a match can easily achieve this at a rough, dry edge like this.
So dry paper works well as a starter fuel. But something more sustaining is needed as pieces of paper alone are likely to burn out before the temperature of the logs has been sufficiently raised. A piece of kindling, much smaller than a log, with its much greater ratio of surface area to volume will offer more surface to ignite and is likely to have dried out more thoroughly for the same reason.

This excursion into the science behind log fires has, hopefully, proved illuminating; but, as any camping enthusiast will testify, actually starting a fire in the open remains more of an art than science!
Happy fire lighting (under controlled conditions and not too often, of course).
© Andrew Morris 2nd May 2020
