
“When a daisy dies, it doesn’t end up as daisy atoms, does it?” So queried Celia in one memorable science discussion, years ago. A lively theatre agent who’d missed out on science at school, Celia found fascinating ways to probe the subject as an adult.
On an autumn walk recently, the same issue cropped up in conversation. What does happen when leaves fall to the ground and seem to gradually disappear?
Figure 1 A woodland walk in Autumn
We have a general understanding that they rot and ultimately become part of the soil. They seem to nourish it in some way, too. But what is actually going on in this process.
Decomposition
When leaves fall from their twigs and branches and settle on the ground they join an active and complex ecosystem, rich in insects, worms and other small creatures but also in a variety of fungi and parasites. Invisible, but no less important, are the many species of bacteria and other microbes that carry out the fundamental chemistry of decomposition. Earthworms, millipedes, slugs and snails start the process off by cutting up the leaf material into smaller chunks, thus increasing the surface area open to the microbes. Studies show that the presence of such animals speeds up the disappearance of leaf material from the soil surface.
Earthworms play a particular role by mixing the layers within soil during their travels through it. This helps integrate new organic matter from leaves into the soil. The Earthworm Society of Britain reminds us that Charles Darwin called them ‘nature’s ploughs’ for this reason. This process is brought to life in a remarkable one-minute speeded-up video showing leaves decaying in the presence of earthworms and other fauna. Bioturbation with and without soil fauna – YouTube. It shows, in just fifteen weeks, leaves are almost completely incorporated into the soil by interacting with animals and microbes.
Fungi
Once the worms, slugs and millipedes have done their work cutting up fallen leaves and churning up the soil on which they’ve landed; what finally does for the leaves to turn them into that soft mulch? Key to this are members of an entirely different kingdom from either animals or plants: the fungi. These ubiquitous organisms exist either as single-celled organisms (called yeasts) or, much more commonly as multicellular organisms. It’s the wall that surrounds each cell that distinguishes a fungus from a plant cell: it is composed of a carbohydrate called chitin which is related to cellulose, of which plant cell walls are made, but is harder and more stable.
Structurally fungi consist of long thin filaments, called hyphae. These are simply a long row of cells linked together and branching off in places. They grow by adding length from the tip of the filament. A single hypha is only a few micrometres in diameter, but many hyphae often bundle together, becoming visible to the naked eye.

Figure 2 Hyphae in a visible bundle

As they branch fungi may form a network known as a mycelium. Figure 3 shows one growing on oak leaves in Ayrshire, Scotland.
A mushroom is the fruiting part of a fungus, rising up above ground. Like other fruits, these play a part in the cycle of sexual reproduction which occurs when two mycelia meet and mate. On its underside, a mushroom contains enormous numbers of spores which act much as seeds do for plants. These can blow away in the wind or be transported by animals, enabling a fungus to start afresh in a different place.
Figure 3 A mycelium growing on oak leaves
The size of mycelia varies enormously: they may be invisibly small or cover hundreds of square kilometres. The hyphae (filaments that make up a mycelium) spread in all directions, branching in all three dimensions, at first. When any of them encounters a source of nutrients they slow down, branch out more and spend time absorbing this food.
When a source is exhausted, the mycelium carries on moving forward, leaving a dead zone where it previously was. In this way an expanding mycelium may advance as a ring, emptying out the space it leaves behind. “Fairy rings” of mushrooms may spring up as the fruiting bodies of the underlying ring of a mycelium.
Figure 4 Fairy ring

Some types of mycelia attach to the roots of plants, enabling a symbiotic relationship to develop. The fungus and the plant both benefit: the fungus supplies water and minerals from the soil for the plant and it, in return, supplies the fungus with sugars that it has synthesised. Sometimes a mycelium may even act as a conduit, enabling chemical signals to pass from one plant to another. Recent research has shown how this may even help plants defend themselves against disease.
We now understand how fungi spread, slowly but relentlessly, throughout soil and dead plant matter, seeking fresh sources of nutrition for themselves. How is it that this process manages to reduce a carpet of fallen leaves (known as leaf litter) to nothing more than an extra sliver of topsoil?
Enzymes
The first stages of decomposition, described above, prepare the ground for the ultimate disappearance of dead leaves. Various creepie crawlies have done their bit, chopping up the leaves into small fragments, exposing much greater surface area to the environment. At the same time, fungi have been extending their tentacles (hyphae) throughout the leaves and soil upon which they rest. Ultimately, for matter to ‘disappear’, it is chemistry rather than biology that does the work. Chemical reactions have to take place to transform the leaf material. How does this happen?
Inside the hyphae (the strings of cells that make up a fungus) are many kinds of enzyme. As explained in blog 2.4 Enzymes, these are giant molecules that act as tools enabling chemical reactions to occur in biological systems. They are capable of cutting up large molecules, like the proteins and sugars that make up food, and also of welding together smaller molecules to make the large ones needed by a specific cell. At the tip of a growing hypha, enzymes are secreted through the wall of the cell into the surroundings. These enzymes act on whatever organic molecules happen to be around.
All sorts of large organic molecules are to be found in the cells of a leaf, dead or alive: sugars, proteins, lipids (fats and oils) and DNA, for example. Enzymes break down these giants into the smaller chemical building-blocks of which they are composed: amino acids and sugars in particular.
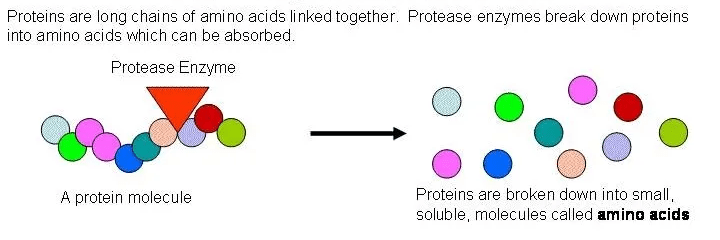
Enzymes called proteases can break the bond between two adjacent subunits in the chain of a protein molecule.
Figure 5 breaking down a protein molecule
Enzymes (called carbohydrases) can break the bond between adjacent subunits in the chain a carbohydrate molecule
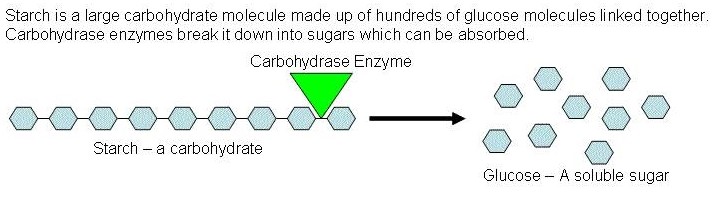
Figure 6 Breaking down a carbohydrate molecule
These are sufficiently small to be simply absorbed through the walls of the growing strand of the fungus. Using energy from the absorbed sugars, the fungus is able to synthesise the proteins and carbohydrates it needs from the fragments obtained from the broken-down molecules of the leaf. Although the proteins in a leaf differ from those in a fungus, the component amino acids of which each is made are identically the same. Similarly, the sugars that make up carbohydrate molecules are the same in leaf and fungus. From the fungus’s point of view, this is how it grows and develops – feeding off the component parts of organic molecules in soil and plant litter.
From the leaf’s point of view, this explains why it gradually disappears. Its once-living structure gets broken down into fragments by small animals, then the cells of which these fragments are composed get disrupted and their contents broken down further by enzymes released from nearby fungus threads

The most digestible fleshy parts of the leaf are tackled by the first wave of enzymes, leaving the tougher, spindly veins for different kinds of enzyme to wrestle with later. This explains the skeletonic appearance of half-decayed leaves (figure 7)
Figure 7 half-decayed leaf, exposing tougher veins.
Soil
As plant material decays, the molecules and atoms of which it is made, are absorbed into the surrounding soil or into the cells of the fungi that grow through it. Ultimately fungi also die leaving behind the organic matter or which they were also made. What happens to this dead organic material once it is taken up in the soil? What indeed is soil?
Molecules, whether organic or inorganic, are composed of atoms. Enzymes in soil are able to break down larger molecules of all kinds into ever small ones – or even into single atoms. Thus material that was once consider organic and may have been part of a living thing, can get broken down into simple substances that enrich the soil. Water and carbon dioxide are abundant products of breakdown; the latter, as a gas simply being lost to the atmosphere. Methane, ammonia, nitrates and nitrites, sulphur dioxide and phosphates are typical of the molecules that remain after organic breakdown; sodium, calcium and magnesium are examples of atoms that remain .
Soil itself is a mixture containing approximately 5% organic matter on average and roughly 45% minerals. The rest is the space between the solid material comprising water and gases. A fertile soil contains the nutrients required to sustain plant life. The carbon, hydrogen and oxygen needed by plants come largely from the atmosphere (CO2) and surroundings (water). The rest comes from the soil and includes phosphorus, potassium, sulphur, calcium and magnesium plus a dozen other elements. These latter derive ultimately from the minerals of which the underlying rocks are made.
Soil thus provides the cocktail of atoms and molecules from which all substances are built, whether in living or inanimate matter. Ultimately, they all originate in the rocks beneath, or atmosphere above, the soil that nurtures the plant and animal life living upon it.
Elements and the cycle of life
Celia launched this blog with a fundamental point about what happens when a daisy dies. As she rightly pointed out, it doesn’t simply disintegrate into daisy atoms. She understood that things are not simply made of out substances peculiar to themselves.

There is some underlying commonality among the infinite diversity of substances. Ancient Greek philosophers had a similar intuition, imagining this underlying commonality to comprise, in the case of Thales, the single elemental substance water; or in the case Empedocles, four elements: air, earth, fire and water.
Figure 8 the four elements of Empedocles
Since the eighteenth century, however, scientists (or natural philosophers, as they were originally called) have devised countless ingenious experiments and made meticulous measurements on different materials. Over time these have shown that all substances are ultimately composed of a limited number of elemental items that cannot be broken down into anything simpler.
All substances are made of some combination of these elements. Thus, for example, water is made of hydrogen and oxygen; ammonia of nitrogen and hydrogen; insulin of carbon, nitrogen, hydrogen, oxygen and sulphur. There are now known to be 98 different elements that occur in nature plus some 20 more that have been artificially created by humans.
Figure 9 Periodic table on a building in Dubna

A daisy is made of cells which are themselves composed of molecules which are themselves made of atoms. Atoms come in the 98 different flavours or elements. There are carbon atoms, magnesium atoms, gold atoms and so on. Ultimately a daisy is made of some enormous number of atoms selected from this range of 98 types. The daisy is unlikely to decompose directly into billions of separate atoms. This is because atoms are attracted to one another, joining together into larger structures – molecules or other kinds of compound structure found in minerals and metals. Thus, a daisy decomposes into large molecules, which break down into small molecules, a few of which may break down further to produce individual atoms.
The key point is that nothing disappears entirely. Some of the detritus of a dead daisy will remain in the soil, adding to its volume, some will be taken up by living things like, fungi and bacteria and gradually reintegrated into living material – perhaps even integrated back into another daisy or the leaf of growing tree. Just a small amount, will drift off as gas into the atmosphere; but even a portion of that might return as new plant life draws in carbon dioxide from the air to sustain the next generation.
Conclusion
By taking note of a simple everyday phenomenon – the decay of fallen leaves – we’ve not only found answers to our immediate questions but also touched on a fundamental issue: the nature the elements. The indestructibility of the atoms and the limited range of their types – the 98 elements – have reminded us of the cycle of life. The decay of one form of living matter creates the conditions for the creation of a new and different form. The atoms of the various elements remain the same throughout; the form into which they are assembled changes.
© Andrew Morris
