Atmosphere, pressure and the middle ear
One evening, deep into a cold November, a fascinating science discussion kicked off, inspired by Julie’s trip to the Canary Islands. Her flight to the sunny south had started badly with a typical British cold: blocked ears and nose. As the plane ascended however, she was delighted to find her ears becoming unblocked. Her sense of relief lasted a pleasant hour or two before she sadly found them blocking up again as the plane descended into Fuerteventura.
She guessed it must have been to do with the pressure in the cabin. This simple suggestion triggered an avalanche of further questions, as the group realised how shaky their grasp was of the basic ideas – What is pressure? What is air? Why do your ears go pop? What about people living high up in the mountains? A great opportunity to explore the nature of the air’s invisible presence.
Cabin pressure
It’s true the pressure of the air inside a plane has to be kept up artificially as a plane ascends because the pressure of the air outside weakens with height. This is achieved by “bleeding” off air that has already been compressed inside the jet engines. If this were not done, air pressure would reduce dramatically inside the plane just as it does when we climb a high mountain. This would leave fewer and fewer oxygen molecules to breathe in. We’d soon get light-headed, as can happen high up a mountainside, and ultimately black out.
But the fact that our ears pop as a plane ascends and descends shows something more: although the pressure inside a plane is kept artificially higher than the pressure outside it is still a bit lower than it is at ground level. This precaution is taken to ensure the difference in pressure between the inside and outside is not too extreme; otherwise there’d be stresses on the structure of the plane and a risk that it could explode. The cabin pressure is maintained at roughly the same level as at the top of a mountain – roughly 2,400 meters, the height of Mexico city. Our ears ‘pop’ when there is a significant difference between the pressure inside and outside the ear – whether it’s in a plane or up a mountainside. ‘Popping’ helps equalise the pressure.
The ear
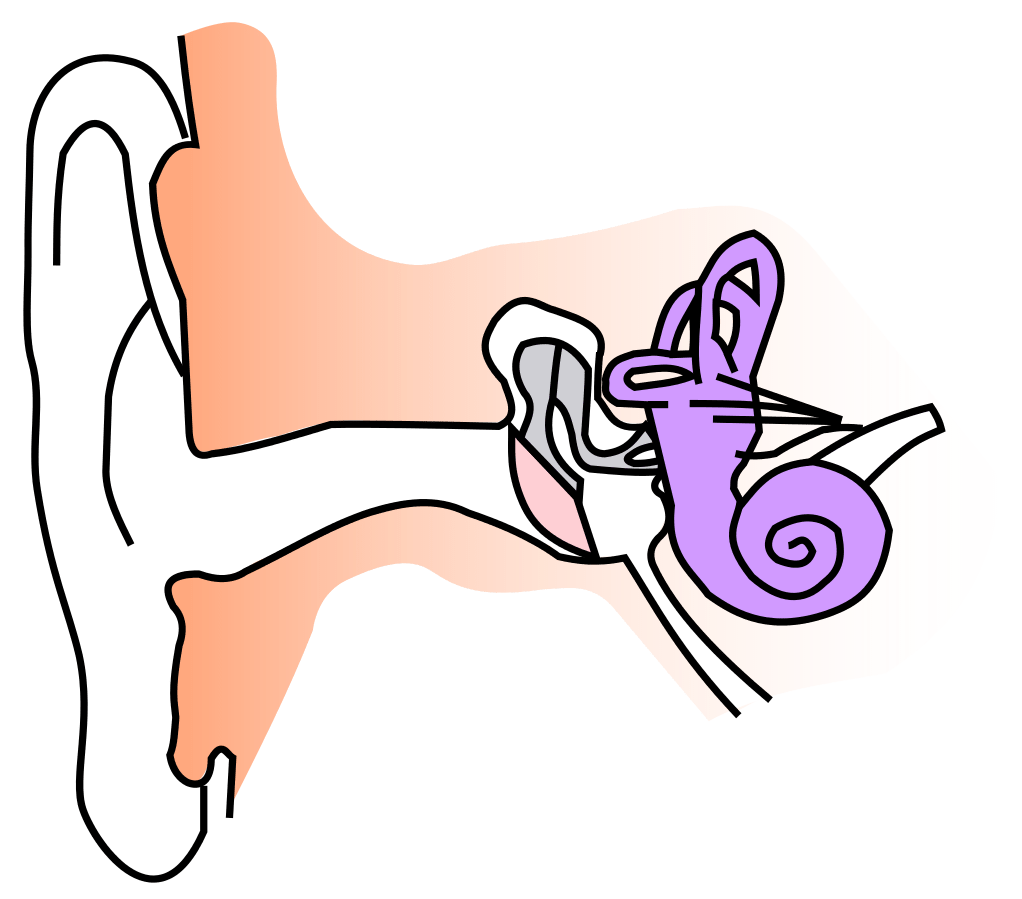
The fact that there’s a ‘drum’ in the ear is pretty well known; but what does this mean? Physically, it’s job is much like that of a musical drumskin: to vibrate. But whereas a musician’s drum is hit by a stick to get it vibrating, the eardrum is set in motion by tiny variations in the pressure of the air surrounding it. These vibrations are what sound waves are made of: tiny fluctuations sent out from the voices, music and traffic that surround us, as this lovely engraving from Popular Science Monthly 1878 illustrates:

It’s hard to believe that such delicate, imperceptible vibrations that you can hardly feel, nevertheless get picked up by the eardrum. In fact the minute changes in air pressure when a sound wave passes is around 2 units (called Pascals) on top of the normal air pressure of 100,000 units. In other words, the eardrum responds to pressure changes of just 1 part in 50,000 or 0.002 %. That’s how sensitive it is.
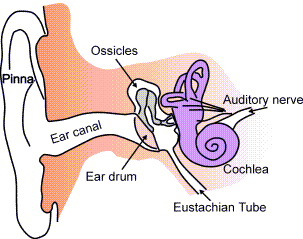
What about the other side of the eardrum? What is it that gets blocked when you have a cold? The middle ear is a chamber containing three miniscule bones (called ossicles) linked to one another. The first of these is attached to the eardrum and together the three of them amplify and transmit vibrations from the outer ear, via the eardrum to the inner ear. It’s here, in the liquid-filled inner ear, that the vibrations stimulate nerve signals to the brain, signifying sound.
Now back to the blocked ear….. the middle ear is a chamber ordinarily filled with air at the pressure of the outside atmosphere. If the external pressure should go down, as it does when an aeroplane climbs high, an imbalance is created with the pre-existing pressure in the middle ear. This gets rectified thanks to a small tube (Eustachian tube) linking the middle ear to the outside world. Normally, this is closed so the middle ear is isolated from the air outside. But when there is a pressure difference across the ear drum it opens up, connecting the inner ear, via the nose and throat, to the atmosphere outside. A bit of air rushes in or out to equalise pressures. A great mechanism for protecting your ear. This is what happens if you yawn or move your jaw when you enter a tunnel in a fast train – you are opening up the Eustachian tube temporarily.
However, this tube can get blocked when you have a cold. It can get inflamed or filled with mucus and fail to open. This 2 minute video is brilliant at explaining it visually. So, somehow, Julie’s blocked-up Eustachian tube must have temporarily opened as the pressure inside her plane reduced from its ground level value, thus relieving her ear drum and restoring normal hearing.
This episode not only sparked a discussion about the working of the ear, but raised further question about what air pressure actually is and why it varies. You can explore these in the following section, if you choose.
Further discussion
Air
Air fascinates us! It comes as shock to us all as children to be told that the apparent emptiness all around us is in fact filled with invisible stuff. It seems absurd at first, and coming to terms with this is an early challenges in our science education. A further shock comes later, when we are told it’s not even a single thing but a mixture of various gases: mainly nitrogen (nearly 80%) with some oxygen (20%) and less than 1% for the rest. But perhaps the most difficult thing for us to accept, even as adults, is that air actually has weight. Grappling with this issue in the group, Sonya asked “if you take a jar of air and a jar of vacuum do they weigh differently?”. Good question – yes they do, but of course, a jar of air weighs so little it would be undetectable with your kitchen scales.
The Pressure of the Atmosphere
Sitting here on the surface of the Earth, however, places us underneath a huge amount of air: there’s about 100km up to the conventional ‘boundary’ between the Earth’s atmosphere and outer space (though there is no boundary in reality – just a gradual thinning out). Even with its very low density, the great column of air above our heads weighs a lot because it is so tall: about 10,000 kg of it sitting above every square meter. That means directly above our bodies (approximately 1/10th of a square meter) is about a tonne of air! That’s why the air around us is pressurised – it’s the air above it pressing down. Fortunately for our wellbeing, the air inside and outside our bodies are both at the same pressure, which explains why our lungs and inner ears don’t explode under the pressure.

The pressure at any given level in any fluid is simply caused by the weight of the fluid above the level. It’s true in water as much as air. This picture shows very directly how the pressure in water gets greater the deeper you go.
Ultimate cause of pressure
At the microscopic level any gas is made of countless millions of molecules rushing around at very high speed. Pressure is simply the net effect of vast numbers of these molecules hitting any given surface. They exert a force on a surface, as a jet of water from a hose does against a wall. The molecules rushing around in the atmosphere hit your body, and, in particular the sensitive part which is your ear drum. They cause no damage under normal conditions because molecules in the inner ear push back with an equal force.
You can see how the idea of molecules rushing around in a gas helps explain simple things we are familiar with. For example: when you pump up tyres you are simply pushing more molecules from the pump into the inner tube. So, more molecules are hitting the walls of the inner tube, increasing the force on it, thus raising the pressure. Another way of increasing pressure is to reduce the amount of space for the molecules to move around in so they hit the walls more often, again increasing the average force on the walls and hence the pressure. This is what happens when you squeeze a balloon. Yet another way to raise the pressure of a gas is to raise its temperature This makes the molecules rush around even faster, and so hit the walls with even greater force. That’s why you shouldn’t leave an aerosol container lying in the sun – it increases the pressure in a confined space which could cause it to explode.
The nature of a gas
The image of a gas as zillions of molecules rushing around sparked an even deeper discussion in the science group. What is in between the molecules in air, or any other gas? asked Mary, quite reasonably. Again, the answer is hard to take on: there is nothing in between the molecules, simply an all-pervasive vacuum. It’s as though something material – air – is simply a lot of emptiness with specks of matter dispersed in it, like dust particles dancing around in a shaft of light. Sonya asked even more philosophically: what is nothing?
This picture of the microscopic nature of a gas – unimaginable numbers of tiny molecules buzzing around at great speed in a vacuum – is one of the fundamental concepts in science. It helps us understand many important things in modern life: how to compress oxygen into cylinders for hospitals; how to design efficient aircraft wings and, of supreme importance now, how to predict changes caused by increasing levels of carbon dioxide in the atmosphere. One more example of how understanding of key concepts in science can help us all make sense of the world in which we live.
© Andrew Morris 22nd January 2020
