
Don’t get me started on glue, said one of our readers, after we’d exchanged initial thoughts about our Pritt sticks. She’d once found herself stuck by the roadside after having made a hash of repairing a damaged wing mirror with superglue.
Her real fascination with adhesives, however, had grown out of admiration for the wonders of glue technology: adhesive strips strong enough to keep a shelf in place and Post-it® notes that stick, yet peel apart. The topic springs to mind in so many everyday situations yet rarely makes it into popular science books or documentaries. Is it really as dull as it at first seems? Superglue was an amazing innovation back in the late 50s and Post-its® a remarkable accidental discovery in the 1970s. But glue has been holding our furniture together for centuries and cement doing much the same for our buildings – and our teeth.
Even more remarkable is adhesion in the absence of glue – daddy-longlegs on bathroom tiles, sheets of glass that refuse to come apart. What is it that makes one substance stick to another? What is it that keeps substances separate from one another in the first place – sand and cement get along fine together until you add water? Come to think of it, why does stuff hold together in the first place? A puff of smoke soon disperses but a lump of coal doesn’t. Let’s start by clarifying a few basics.
Materials
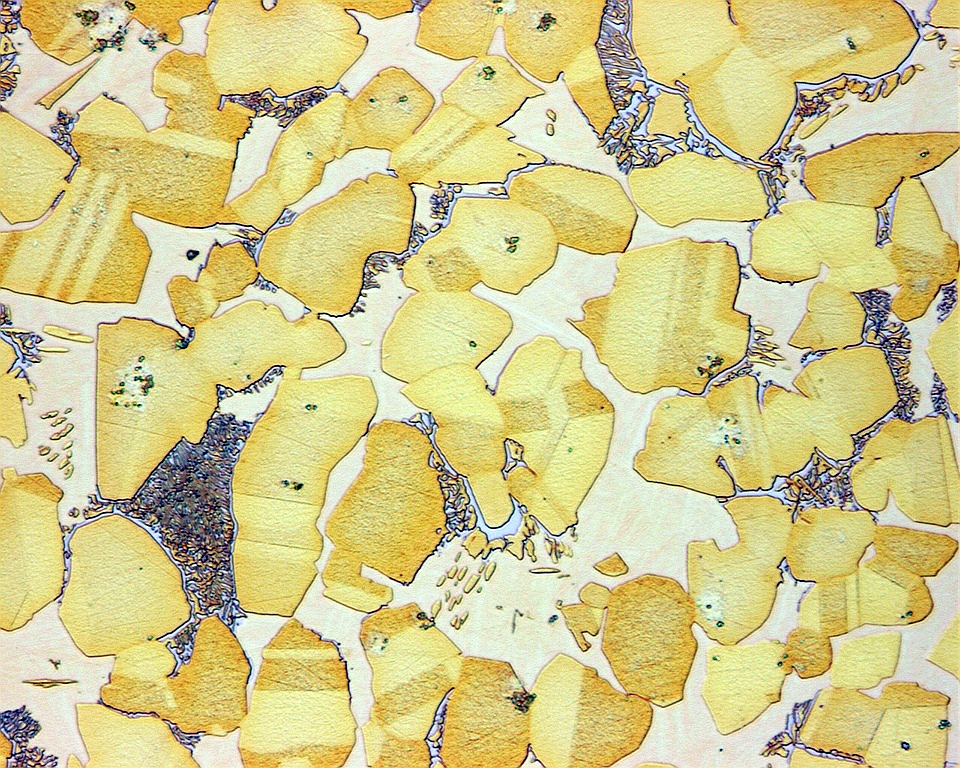
Most of the materials we come into contact with are solid – metal, plastic, cement, wood. If you were to look at them in detail through a microscope you’d see they are each made up of microscopic structures at a more granular level. Metals, for example, consist of grains, as this microscope photograph of grains in Aluminium – Bronze alloy shows.
Image credit: Eisenbeisser
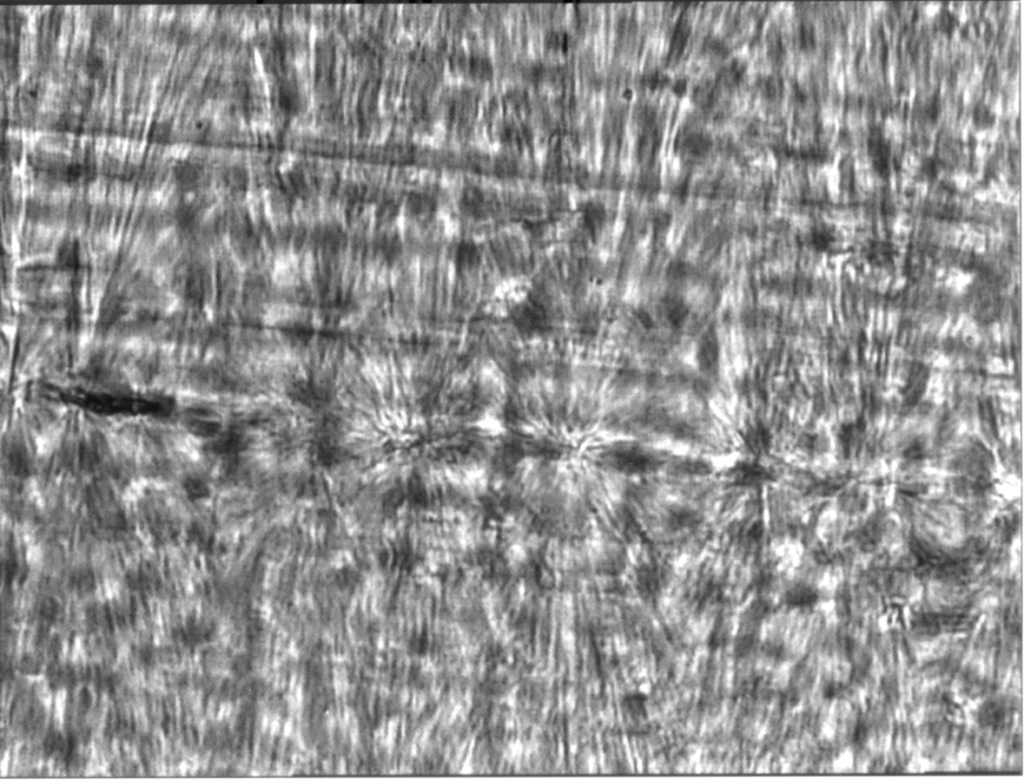
Plastics are made of polymer molecules – that is long chains of repeating links. These come together to form regions that are semi-crystalline, known as spherulites. This photograph of polypropylene, taken through a microscope shows these semi- crystalline structures.

Cement used in building is also composed of grains when seen under a microscope. The photo on the right is a limestone based cement.
Image credit: Zhijun Tan (土木坛子)

A different kind of cement is used to hold bridges and crowns in place in dentistry. Though made of different substances , it also has a granular structure, as shown in this microscope image .
Credit: Journal of Biomedical Science and Engineering
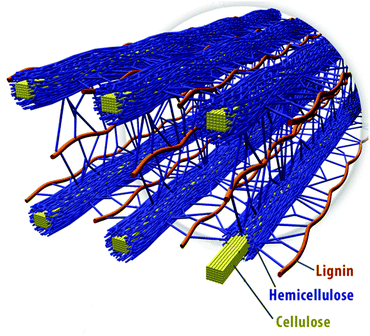
Wood, on the other hand, is made of fibres at the micro level. Long thin molecules of a protein called lignin line up with microfibres of hemicellulose and cellulose with which they interconnect.
What each of these materials have in common, whether granular, crystalline or fibrous, is that their interiors have an irregular structure which leaves their surfaces rough at the microscopic level. As a result when they come into contact with one another, they are less intimately interconnected than appears to the naked eye.
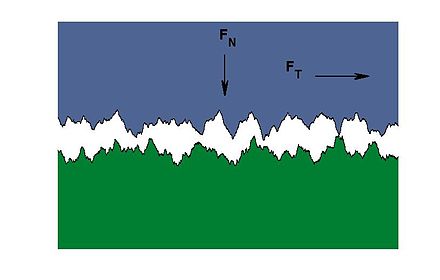
This diagram gives an impression of how it might look at the microscopic level. The jagged surface of the blue object sits irregularly on the jagged surface of the green one. It’s only at very few points that the surfaces physically touch each other.
Image credit : CaoHao of Matlab
Incidentally this image also shows why it takes some force (FT) to drag one material across the jagged surface of another one – what we know as friction. Imagine those peak and toughs grating as they pass each other. The arrow FN also indicates that, if we press one object down on top of another, the jagged surfaces mesh more tightly together, making it harder to slide one over the other – increased friction.
The role of adhesives is made immediately apparent by this diagram: they are needed to fill the space between two surfaces, to connect them much more intimately.
Adhesives
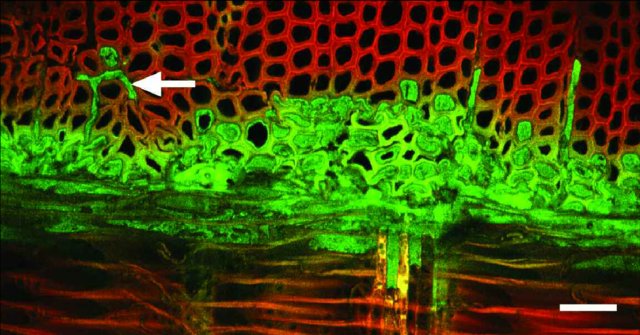
This microscope photograph shows beautifully how an adhesive, in liquid form, penetrates and fills the gap between two surfaces. It was taken by Bernard Dawson, a scientist at Scion, the government forestry research institute in New Zealand.
It shows a line of adhesive (green) between two sheets (or plies) of wood (red). The adhesive can be seen penetrating deeply into the cellular structure of the wood on either side. To indicate the scale, the white line at bottom right represents 50 micrometres (millionths of a metre).
Adhesives work their way between surfaces to perform their binding role in a variety of ways. This neat diagram by Chris Woodford of explainthatstuff.com illustrates them.
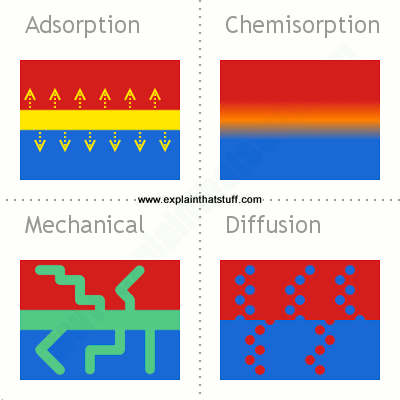
In the simplest case, molecules in the adhesive and the two surfaces may simply attract one another – adsorption. Alternatively molecules in the adhesive may react chemically with those on the two surfaces – Chemisorption. In other cases, molecules may cross between the materials (diffusion) or penetrate the cracks in them, linking them physically (mechanical adhesion).
It’s clear from each of these images that the adhesive needs to spread evenly throughout the gap between the two surfaces – it needs to “wet” them. This fits with our everyday experience of glues as liquids, gels or pastes. But to become firm and fast after having wet the surfaces, they also need to dry out over time. To achieve this the adhesive is often combined with a solvent that evaporates, such as water, acetone or ether – that’s the smell of glue setting. As well as sticking firmly to other materials, glues also need to hold together strongly within themselves, so that the join doesn’t come apart within the glue itself. In other words it must cohere as well as adhere.
The attractive force
Filling the space between two surfaces and perhaps penetrating into them is clearly necessary for glues to work, but what is it that actually does the sticking ?
The explanation lies in a simple but fundamental aspect of all substances: the electrical nature of atoms. It had long been thought that all the diverse substances we see are made up of tiny, fundamental components ,to which the ancient Greeks gave the name “atoms”. What was not suspected till atoms were actually discovered, just over a century ago, was that these are electrical in nature. This seems to be counterintuitive as they appear to be perfectly neutral – neither positively nor negatively charged. The surprise was to find that atoms are not the end of the matter: they are themselves made of smaller parts and these are electrically charged. The trick is that the positive and negative particles inside atoms are exactly balanced. That’s how they come to be neutral, normally. For a more detailed explanation see the associated blog: Inside the atom.
For adhesive substances it’s the electrical attraction between positive and negative charges that accounts for the bonding. Molecules are made of atoms and, like most substances, glues are made of molecules. Inside some molecules one part can be a bit more negative than another, leaving other parts a bit more positive. Where a molecule like this meets another such molecule the positive and negative regions (or poles) can be attracted to one another, much as two magnets are.
These forces of attraction between two molecules are pretty weak in themselves but add up when many molecules come close to one another. In the interior of substances molecules are close to one another (except in gases). This is what keeps them together. When two objects come into contact, however, few molecules in the objects come close due to the roughness of the surfaces. That’s why most substances don’t ordinarily stick to one another. Adhesives however fill up the gaps and ensure that the molecules of which they are made get right up close to those in the material they are sticking to.
The special gift of an adhesive is to bring large numbers of molecules in two different substances very close together. The strong bond that develops is the result of a huge number of tiny forces adding together.
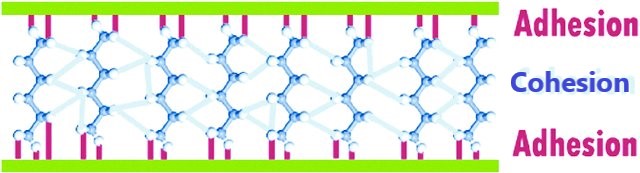
Glue molecules have to be attracted to molecules in the two different substances (adhesion) but also to one another (cohesion), to make sure the glue itself doesn’t split apart.
Examples
Now we understand the principles, what about the practice? Glues and cements appear in all sorts of situations, from bookbinding to dentistry, wall paint to insect feet. Some examples from everyday life are explored in this final section.
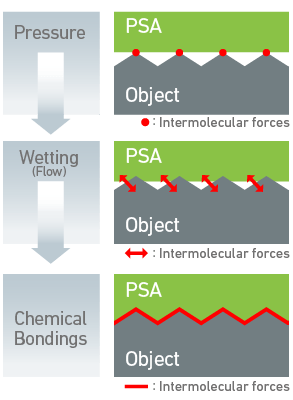
Sticky tape
Formally known as pressure sensitive adhesive (PSA), sticky tape combines a viscous adhesive (often a polymer) with a plastic film on which it is fixed. The backing of the film has to be coated with another substance to prevent the tape from sticking to itself.
Pressure from your hand helps the adhesive flow and allows the tape to make closer contact with the surface, increasing the amount of force between the molecules. If dust or oils gets in between it reduces the contact area, weakening the bond .
Image courtesy of DIC Corporation, Tokyo
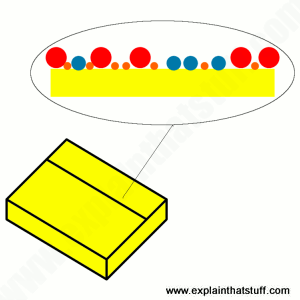
Post-it® notes are known as “low-tack” pressure-sensitive adhesives. This means they do not wet or run rapidly like other glues. The sticky part contains tiny bubbles of adhesive, so when pressed onto a surface there’s just enough adhesive force to grip lightly.
At first the 3M corporation considered the accidental discovery of a new weak glue to be a failure. Years later an employee started using it to fix bookmarks in a book; some market testing soon led on to commercial success. Apparently the lab only had yellow paper to test it on – the colour has stuck.

Glue sticks
This innovative type of glue was developed in 1967 by researchers at Henkel in Düsseldorf. The idea of a glue stick was a departure from the usual liquid type. To get the glue to stiffen a soap gel was combined with the water-soluble adhesive, making it solid. When rubbed on paper the adhesive components are released creating an adhesive film. Lipstick was the inspiration for the twist-action. Apparently the name for the original brand came from the inventor’s child mispronouncing “pretty stick”.
Superglue
Formally known as cyanoacrylate, superglue works differently from most other glues. Instead of using a solvent that evaporates to leave a firm bond, the molecules of cyanoacrylate rapidly fuse together in the presence of water, forming long chains (or polymers). Even the water in the atmosphere or on your skin is enough to set this off. The polymers then bond strongly to the molecules in the surfaces to be glued.
The discovery of superglue was another example of serendipity. It began during the second world war in an effort to find a clear plastic for gun sights. The research was abandoned when the substance turned out to be annoyingly sticky. It was accidentally rediscovered later by researchers at Eastman Kodak who foresaw its commercial potential.
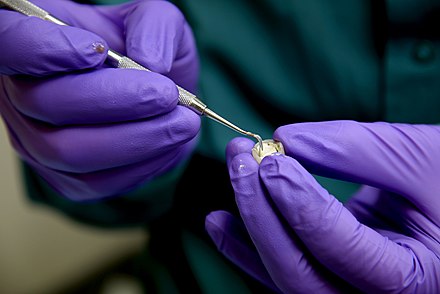
Dental cement
As with superglue, dental (or ‘luting’) cement, does not involve a solvent drying off. Instead a chemical reaction takes place between a dry and a wet ingredient. As the cement hardens it bonds with the two surfaces. Sometime halogen or LED light is used as a catalyst to speed up the reaction.
This image is of a crown to which cement is being applied.
Cells
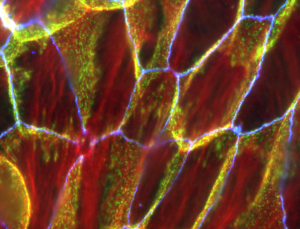
A fundamental kind of adhesion common to all living things (apart from single cell organisms) is the stickiness that holds our living cells together. Without this we’d just be a heap of powder rather than the complex tissues our organs and connective tissues are made of. Adhesive molecules interact between the cells (blue lines)
Image credit: Northwestern University
The ‘glue’ that connects cells comprises adhesive molecules that create junctions between cells. They are not only tight – so tissues hold together even under strain – but also extremely flexible, as we know every time we stretch or even breathe.
An adhesive molecule (called cadherin) links through a protein to the internal structure of a cell. It’s recently been discovered that these proteins are capable of assembling, disassembling and reassembling thousands of times in a second. It’s this rapidity with which connections between cells can be made and broken that enables our tissues to be flexible yet hold together firmly.

Geckos
Geckos are able run around upside down thanks to thousands of tiny hair-like bristles on the underside of their feet. Even smaller projections are attached to these. As we’ve seen with all other adhesives it’s this multiplication thousands of times that creates sufficient attraction to hold surfaces together.
Geckos, like post-it notes, need to also detach their grip rapidly as they move. The bonds between the hairs on their feet and the surfaces on which they scamper are easily broken.
Image credit: Bjørn Christian Tørrissen

The gecko’s anatomy inspired scientists at Stanford university to develop a device enabling humans to climb a vertical glass surface. By assembling thousands of microscopic wedges of silicon rubber onto tiny tiles set onto pads they were able to generate adhesive forces just as the gecko does.
Image courtesy of Biomimetics and Dexterous Manipulation Lab, Stanford University
Conclusion
What strikes me as remarkable after looking into glues, is the simple and unifying principle that underlies the diversity of adhesive types. It’s hard to imagine that the glue holding the tiles on a returning space capsule works on the same fundamental principle as that on the back of a Post-it® note. Of course the formulation differs but the underlying mechanism remains the same: attractive forces between molecules. Weak as they are individually, we hardly notice them in ordinary circumstances. Plates don’t stick to tables, handshakes are easily released. The job of any adhesive is to increase dramatically the number of molecules in close contact between two surfaces so these tiny forces multiply up to a significant grip.
A simple exploration of everyday objects leads us once again into one of the fundamental concepts in science.
©Andrew Morris 28th March 2021
