With the pandemic in full swing globally, immunity is the great hope of humanity. This wondrous condition promises to free us from the ravages of the virus, paving the way to the life we once knew. But what exactly is it, how do we attain it? Is it permanent, will it survive the onslaught of the variants?
With change happening at such a pace, today’s information will be out of date tomorrow. Scientific research can only track the evolution of the virus, not predict it. These urgent questions can only be answered in real time as studies assess the effects of the unfolding disease. It’s through the daily news bulletins, not the text book, that we’re learning our science now. Here we explore what is known about immunity and it’s limitations, to help us interpret the news that’s yet to come.
Immune system
It’s a system – that’s the first striking feature of our defence apparatus. It’s not a discrete organ or chemical that fights infection. It’s a system that has evolved gradually over the aeons, as simple organisms gave way to sea, then land, creatures and ultimately higher mammals. Sea sponges are able to distinguish grafted tissue from their own, while higher up the evolutionary tree we find cells capable of engulfing and destroying unwanted material. Higher still, insects have substances that attach to unwanted cells, causing them to aggregate and antibodies appeared with the arrival of vertebrates. The complex system we humans have inherited – and share with reptiles, birds and mammals – involves elements of each of these ancient defence strategies.
The result is a set of distinctive bodily responses that protect us against a huge range of threats from viruses, bacteria, parasites and a host of other microbial beasties. Together, they are capable of recognising and ridding our bodies of whatever doesn’t belong.
The first line of defence of the system is the physical barrier presented by tissues such as skin and mucus to external substances, which prevent the entry of many unwanted bugs (or pathogens to be more precise): some however have evolved to get through this. Cuts and fissures and the openings of the respiratory tract are well-known entry points.
For those that manage to get through, the next line of defence they encounter is the innate immune system. As its name implies, this lies ready and waiting at all times. It responds rapidly to a broad range of the more common microorganisms. Molecules on the surface of familiar pathogens are recognised by the system which then mounts an inflammatory response: redness, swelling, pain and temperature rise. These well-known symptoms of infection are caused by the release of various kinds of chemical which dilate blood vessels, activate pain receptors and attract defensive cells to neutralise or destroy the pathogen.
Some pathogens, particularly unfamiliar ones, such as the present coronavirus, manage to get past this line of defence. The next level of protection is provided by the adaptive part of the immune system. As its name implies this adapts to whatever arrives – it copes with the unfamiliar.
Two distinct kinds of white blood cell form a major part of the adaptive immune system. One type, known as B cells, produce antibodies targeted at the specific unfamiliar pathogen that has entered the body. These move through the bloodstream to wherever the alien particles are and lock on to them, inactivating them. The other type of white cells, known as T cells, have several functions, including killing alien cells and helping B cells produce antibodies. Remarkably, both B and T type cells develop in such a way that they become specific to a particular pathogen, like the coronavirus, for example. They recognise a particular antigen – such as the spike protein on the surface of the virus – and selectively produce antibodies that interact with it. The system adapts itself to cope with the new, unfamiliar threat.
Even more remarkably, a number of these B and T cells help prevent reinfection. They retain the specific antibodies to the particular pathogen, keeping them in abeyance, ready to resist immediately any future occurrence. In effect the system remembers previous encounters with the pathogen. New born babies, of course, have no such “memory”. To protect them, antibodies from the mother are passed through the placenta and milk which last until the baby develops its own during its first few months
Specificity
Perhaps the most remarkable feature of the system is its ability to recognise almost any horrible bug that’s thrown at it: measles, mumps, colds or flu. Like so many recognition processes in the body, this is thanks to the way protein molecules interact with other smaller structures.
Proteins of the immune system are large globular molecules with knobbly surfaces pitted with crevices. They have evolved in such a way that particular sites on their surfaces fit neatly around particular small molecules with which they interact. They are as locks to the keys presented by the smaller molecule (or part thereof). This geometrical ‘fit’ between the larger protein and smaller molecule is enhanced by electrical attraction between negative and positively charged zones around the atoms of each molecule.
Protein molecule (blue) interacting with a small molecule (red)
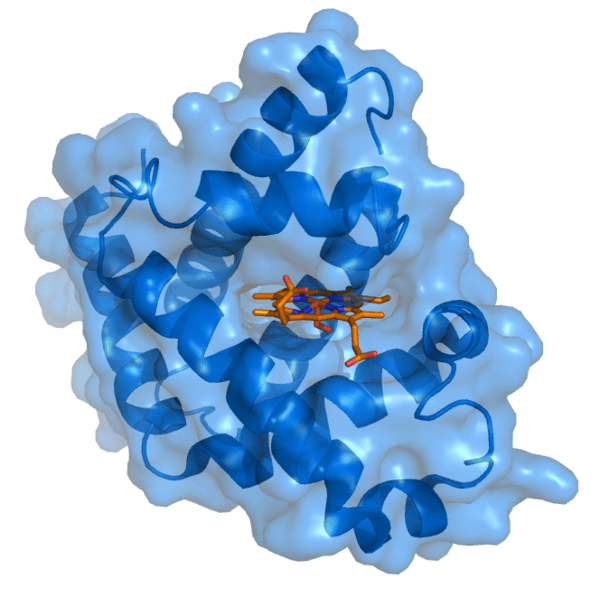
It’s this fit that underlies the great range of interactions that keep our bodies functioning: enzymes snipping up carbohydrates in our intestines, oxygen latching on to haemoglobin in the lungs and antibodies singling out unwanted pathogens, such as coronaviruses, for example.
Antibodies
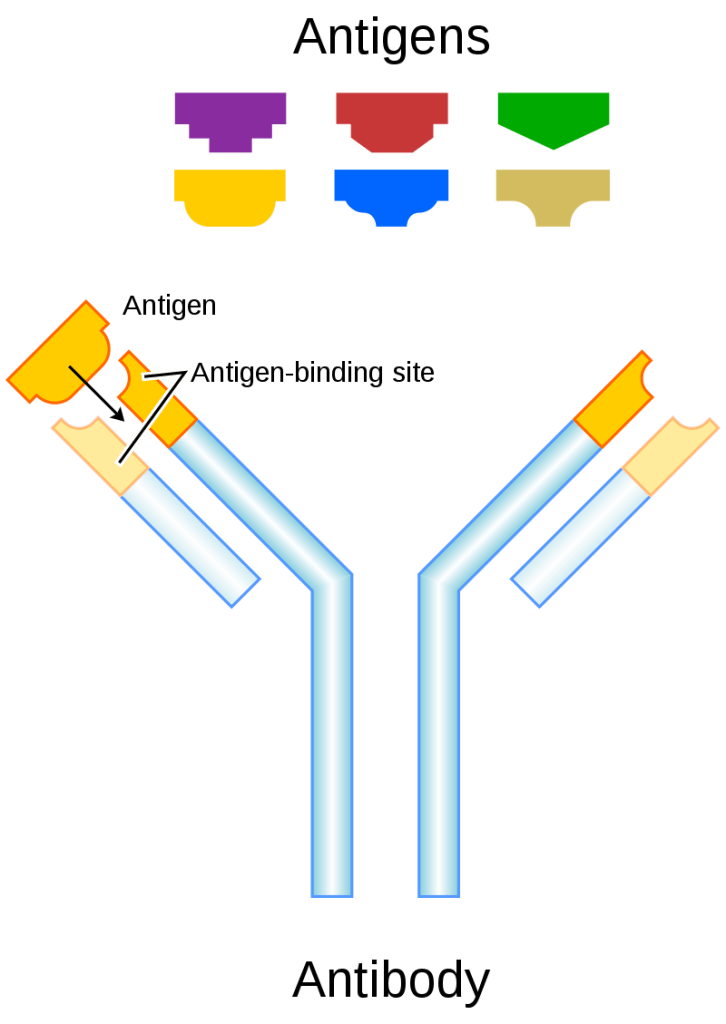
Antibodies are an extraordinary type of giant protein molecule. Like other types they have their special sites, their crevices, shaped to fit the smaller molecules to which they must attach. But unlike other proteins they must be capable of ‘recognising’, and binding to, smaller molecules (or parts of big molecules) of a vast number of different kinds – bits projecting out from all the various unwanted types of bacteria, viruses, parasites and fungi, for example.
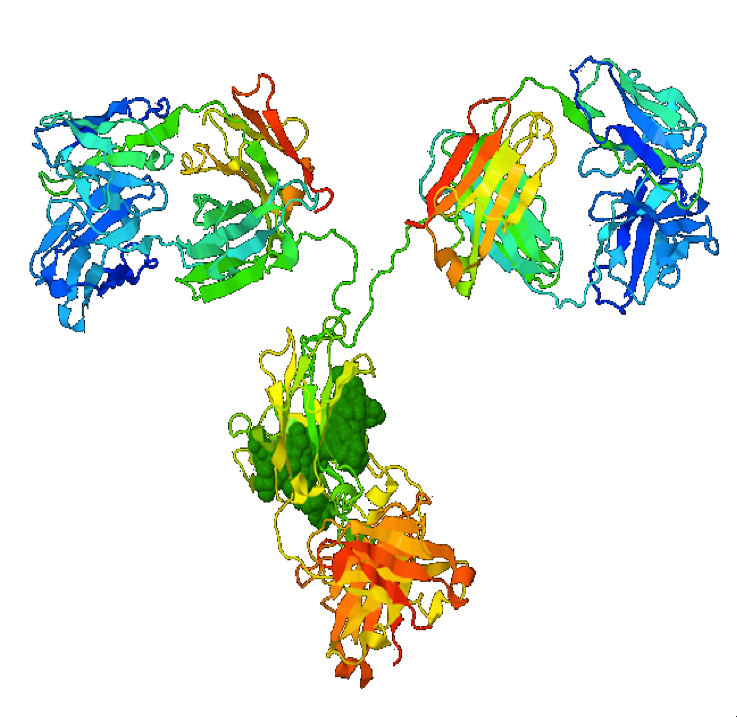
To achieve this enormous flexibility, a unique architecture has evolved within the family of antibody molecules (known technically as immunoglobulins).
In the model on the left each little blob represents an atom. Linked together, the many thousands of atoms constitute the giant Y-shaped antibody molecule. Two chains of atoms (blue and yellow) run from top to bottom. A narrow hinge region in the middle confers enormous flexibility to the molecule around its waist. This is shown even more clearly in the image at the top of this blog.
Two further chunks (green and pink) form part of the upper two arms of the Y shaped molecule.
Image courtesy of Tokenzero
Antibody molecule (or immunoglobulin)
At the tip of each arm of the Y is a section of the molecule that has the remarkable ability to change shape. Called the binding site it is able to adapt its shape to fit any of the countless pathogens to which it has been exposed.
To explore this unique property of antibody molecules see the Further Information page.
Natural immunity
It’s the extraordinary chemical memory of the system that accounts for the well-known experience of immunity after previous exposure to diseases like mumps or measles. Antibodies and white cells remain stored in the body for long periods of time – up to a lifetime in some cases. If the pathogen were to reappear at a later date these specific memory cells of the immune system would recognise it and multiply rapidly to eliminate it.
It’s common experience that it doesn’t always work as neatly as this. The degree of immunity can wane over time and, as we know only too well from seasonal influenza, a virus can mutate, becoming unrecognisable to the memory cells. In the case of the coronavirus responsible for COVID-19, the medium and long term outlook are still unknown. The virus is indeed mutating and cases have been reported of patients infected for a second time. In the case of the common cold, it is still not understood why natural immunity seems to be relatively short lived.
Vaccination
Immunity can also be induced artificially – that’s what vaccination is all about. The vaccine causes the antigen – a fragment of the spike protein in the case of coronavirus – to appear in the bloodstream. This stimulates the immune system to mount a defence, just as it would do if the infection had occurred naturally. It develops specific B cells and T cells and antibodies capable of responding to the new pathogen. These multiply as they would for a natural infection and ultimately destroy the material inserted via the vaccine . Crucially, copies of the specific B and T cells are retained as a kind of ‘archive’. As a result, any encounter with the virus at a future date calls up the ‘memory’ of the antigen from the vaccination, enabling a full defence to be rapidly mounted based on the copies of the cells and antibodies in the archive.
In the case of coronavirus, different vaccines work in different ways. The most well-known ones do not in fact contain the spike protein or any other antigen. Instead, they contain the genetic code (RNA) for making the spike protein and this is injected into the body inside a protective coat. This finds its way inside our cells where it gets translated by the normal machinery of the cell to produce large numbers of copies of the spike protein. These copies, made inside our bodies, then go on to stimulate our immune systems.
Click to see a beautiful model of a Covid-19 vaccine. Details of various types of these vaccines are given in the Further Information page.
Mutations and variants
As we’ve seen above, the proteins of the immune system are giant, globular-shaped molecules.
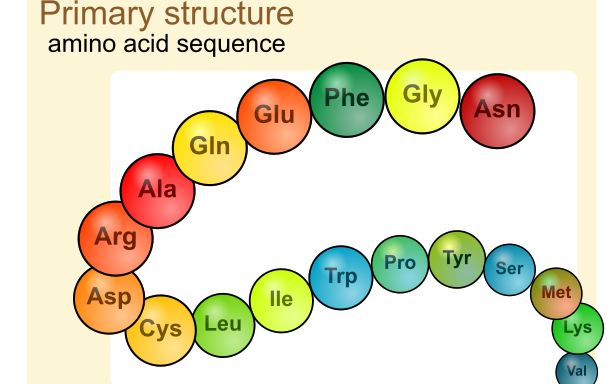
The apparent shape is deceptive because the molecules are long thin thread-like structures, that wrap up into a globule – like a beaded necklace held in the palm of the hand. The thread is a sequence of subunits linked together like the beads in a necklace. The exact sequence of subunits in a given protein is determined by information coded in the gene upon which they are based.
This is how living systems normally function – proteins are continuously being made using the genes encoded in DNA or RNA; and every time a cell divides in two its DNA/RNA gets duplicated.
But very occasionally mistakes occur as genes are duplicated for transmission from one generation to the next. One of the ‘letters’ in the genetic code may be misread; or a random collision with a high energy particle (in cosmic radiation or X rays, for example) – may replace one ‘letter’ with another. This is a mutation – from the Latin word for alteration. If the code is changed, the protein which is based upon it may also be changed. Fortunately for us, when such changes – or mutations – do occur, they usually make little or no difference to the behaviour of the protein. Just occasionally however they do.
Once a gene has been altered, whether by miscopying or radiation effects, the alteration gets reproduced when the gene replicates. In larger scale organisms with long lifecycles, such as us humans, this transfer of a mutation from one generation to the next will only occur once every few years or decades. Viruses, on the other hand, bring forth the next generation in a matter of minutes. Many generations are passed through in the course of a day and each occasion introduces the chance of a mutation. That’s why it’s so important to bring down the overall amount of virus in a population as quickly as possible, to minimise the number of replications, each with its attendant risk of a mutation.
As with evolution generally, different versions of a virus will survive and reproduce to different degrees. Those that survive longest and produce the largest number of offspring, will outnumber the others in the population. With such rapid turnover, new versions of the virus that are well fitted to their environment will soon spread amongst the general population of viruses.
Over time the genetic material of a virus will acquire more than one mutation at various points along its sequence of thirty thousand bases (or ‘letters’). The word ‘variant’ is usually used to denote a version of the virus containing several mutations. If a variant becomes established and starts behaving in a distinctly different way it may come to be called a ‘strain’ (though the terminology is not precise).
At the time of writing thousands of distinct mutations have been identified – i.e. locations on the viral RNA where a base (i.e. a single letter in the code) has been altered. This is entirely normal and the vast majority cause no harm. The number of variants – each with its characteristic set of mutations – that are viable and able to spread is much fewer. The effects they each have on disease symptoms is being closely monitored as and when they are identified. The colourful chart below, maintained on a daily basis by the international open-source collaboration Nextstrain, shows each ‘clade’ (virus family descended from a common ancestor) in a particular colour. Each blob represent a unique genetic sequence identified in a laboratory somewhere in the world. The number of mutations in each case is indicated along the horizontal axis. There are hundreds of thousands of these slightly differing sequences. Because the geographical location at which each mutation was first identified is recorded and shared globally, the journey made by each specific variant as it crosses the world can be plotted. This is how we are able to trace patterns of transmission across the world.
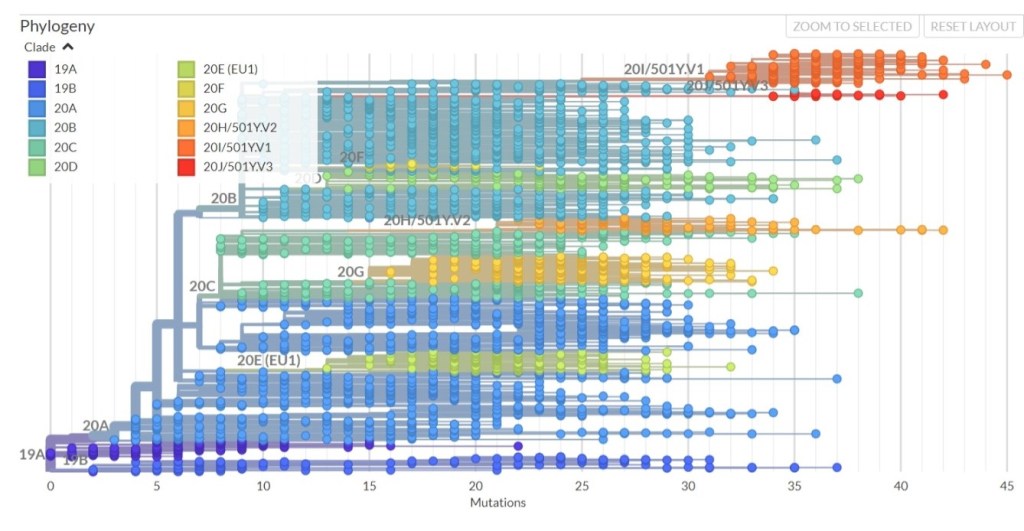
Genetic sequences of coronaviruses, organised by family
The ability of vaccines to respond to each of the variants is a matter of continuous research in real time around the world. Fortunately the latest method of delivering vaccines, by enclosing the gene for the spike protein in a particle that can enter our cells, should help cope with these variants. It means that the gene for a new variant can be inserted into the original, already-tested delivery particle, thus speeding up the time taken to update the vaccine.
The future path of the pandemic is, of course unknowable. All of us are on the same footing now, as we wait on news from the epidemiology and virology labs. With this grounding in the basic science under our belts, perhaps the news will seem a little more intelligible whenever it comes.
© Andrew Morris 6th March 2021
Click to receive an email alert when each new blog is published.
