Atoms and elements are words known to many but understood by few. They crop up in discussions everywhere from medical treatments and aircraft manufacturing to the depths of outer space. The difficulty many of us have is distinguishing between the two and grasping what each word really means.
It’s not hard to grasp the difference and it clears up a lot of confusion if you do. Both words concern the variety of the substance of which the world is made. The basic question we all ask at some point is “what do you end up with when you break things down repeatedly?” Is there one fundamental building block or are there many. The answer, it turns out, is slightly more complicated than you’d hope. But one thing is very clear: you don’t have different building blocks for each separate thing – no grass atoms, cement atoms or wood atoms. There are much fewer types than that.
 The first thing to became clear concerned the nature of different substances. Later, the structure of them became clearer too. It was gradually revealed that some substances you encounter are just themselves under all conditions and can’t be broken down into more fundamental substances – they are elemental and hence called ‘elements’. There are beautifully displayed in the Periodic Table. Most substance are not like this – they are made from more than one element and are called compounds.
The first thing to became clear concerned the nature of different substances. Later, the structure of them became clearer too. It was gradually revealed that some substances you encounter are just themselves under all conditions and can’t be broken down into more fundamental substances – they are elemental and hence called ‘elements’. There are beautifully displayed in the Periodic Table. Most substance are not like this – they are made from more than one element and are called compounds.
The second key concept is the atom: the fundamental structural unit of each element.
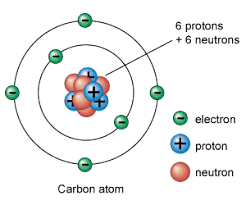 The important point is that all atoms have the same overall architecture; they are tiny and round and are all made of the same fundamental components – various kinds of particle. What distinguishes the atom of one element from another – gold from carbon or sulphur, for example, is not the things of which they are made, but how many of these particles there are and how they are arranged.
The important point is that all atoms have the same overall architecture; they are tiny and round and are all made of the same fundamental components – various kinds of particle. What distinguishes the atom of one element from another – gold from carbon or sulphur, for example, is not the things of which they are made, but how many of these particles there are and how they are arranged.
Atoms of various elements make up everything we know (almost) and help us understand all matter – from bricks and mortar to flesh and bones. Experimental evidence gradually showed that more than one atom was involved in the building blocks of many substances. A number of atoms joined together, as in water H2O or carbon dioxide CO2, are called a molecule.
Longer version of this blog with more detail.
