Cholesterol, oestrogen, carbohydrate, nicotine, protein, styrofoam, perspex … names that crop up in everyday language today; but what exactly do they refer to? What makes up these substances? What distinguishes them from each other; what have they in common? This is what we’ll deal with in the next ten minutes.
All of these substances – and almost any other you can think of – are examples of compounds; that is, they are made up of more than one basic thing. They are made up of atoms stuck together and these atoms are of more than one element. Take carbon dioxide for example: it’s a compound of one atom of the element carbon and two atoms of the element oxygen. Water, H2O, is a compound of two hydrogen and one oxygen atoms. It’s that simple.
Some compounds are more complex, of course. Cholesterol, for example, contains 27 carbon atoms, 46 hydrogen atoms and one oxygen. That’s why it is known technically as C₂₇H₄₆O. Others are much larger, some are gigantic. A typical protein, for example, may comprise tens, or even hundreds, of thousands of atoms; a strand of DNA may contain billions.
Thanks to painstaking experiments, beginning in the eighteenth century and continuing to this day, we can go further than this. We can talk not just about the composition of substances – what they are made of – but also about the structure at the fundamental level. We can say how the atoms in a compound are attached to one another. It’s rather like being able to say not just that a house is made of 7354 bricks, 94 planks and 350 tiles but that these items are connected up in such and such a way to make this particular house.
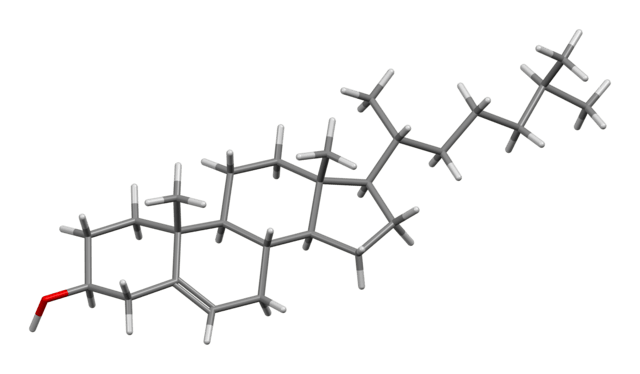
The basic structure of all the substances mentioned so far in this piece, is the ‘molecule’. This is simply a group of atoms connected together (more technically: ‘bonded’). Thus, a sample of cholesterol – a compound of carbon, hydrogen and oxygen atoms – comprises zillions of cholesterol molecules, and in each of these, the atoms are bonded to one another as in the images below. These two models of a cholesterol molecule are each designed to reveal how the molecule is structured. The first emphasises the connections between the atoms (the ‘bonds’) and the second the amount of space taken up by the atoms. In both models the 46 hydrogen atoms are coloured white, the 27 carbon atoms black, and the single oxygen atom, red.
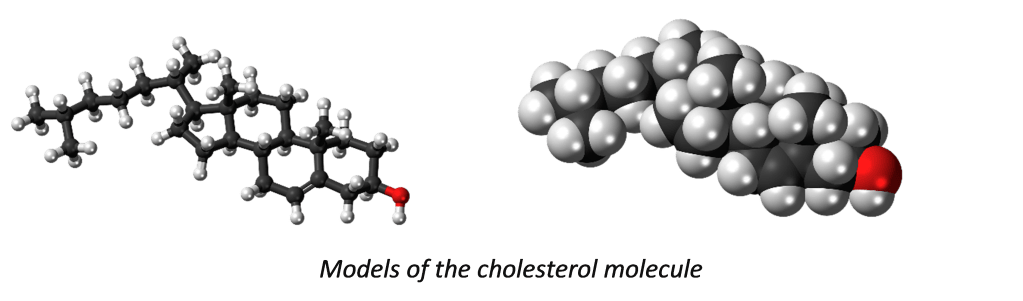
A molecule is thus a group of atoms bonded together. The molecule is the smallest unit of structure for many of the compounds we come across in everyday life. But the size of the molecule varies from substance to substance. The cholesterol ones above are medium size. A very small one is the water molecule and large one is the protein, insulin.

Most of the substances we come across in everyday life are made up of molecules. Compounds associated with living things – like food, leather and flesh – are made of molecules based mainly upon the element carbon. These are the subject matter of organic chemistry. Many so-called plastics are also made of molecules based on carbon, usually in the form of long chain-like molecules known as polymers. These also play an important part in organic chemistry.

However not all substances are made of molecules. Common salt and chalk, for example are not, and the same goes for many substances we generally come across as minerals. These are the subject of inorganic chemistry.
The atoms that make up these substances are not linked together into discrete molecules. Instead they are normally held together in a kind of three-dimensional lattice, or crystal. The diagram shows the compound sodium chloride (common salt); sodium atoms are shown in purple and chloride ones in green.
Within each of these atoms, one or more of the negative particles (electrons) gets transferred from one kind of atom to the other. This occurs because the overall arrangement of electrons achieves a state of lower energy in this way. In the case of common salt (sodium chloride), for example, an electron is transferred from each sodium atom to each chloride one. As a consequence the chloride atoms become negatively charged because they have acquired an extra electron. Similarly, the sodium atoms are positively charged as they have lost an electron. These charged versions of an atom are known as ‘ions’. It’s because these ions have opposite charges that they attract each other, thereby sticking together to make a solid compound: crystals of salt instead of separate sodium and chlorine atoms.
In another blog, we’ll see what goes on inside atoms. This will help us see how they join together to make molecules or sit as neighbours in a crystalline array.
© Andrew Morris 5th November 2021
